COVID-19 vaccines
RT: A small European country defeats coronavirus because it is not a member of the EU
It is about San Marino, and its victory over the coronavirus can be attributed to the fact that it is not a member of the EU, so it did not have to follow the strict rules of the regulatory bodies of the Union about which vaccines we can use, writes Russia Today.
Turkey lowers jab eligibility age to 16
In an effort to further increase the coverage of its inoculation program, Turkey has lowered the eligibility age for the vaccine against COVID-19 to 16.
Those aged 16 and over are now entitled to make appointments to receive their first doses of the jab.
- Read more about Turkey lowers jab eligibility age to 16
- Log in to post comments
WHO sees ’likely’ Covid vaccine link to rare heart inflammation
The World Health Organization said on July 9 that there was a "likely causal association" between coronavirus vaccines using mRNA technology and "very rare" heart inflammations, but the benefits still outweigh the risks.
CoronaVac vaccine 83.5 percent effective: Turkish study
Interim results from a phase 3 trial in Turkey have shown that two doses of CoronaVac, a vaccine developed by the Chinese firm Sinovac, are 83.5 percent effective against symptomatic COVID-19.
COVID-19 vaccination campaign: 16,183 people immunised in 24h
Romania's National COVID-19 Vaccination Coordination Committee (CNCAV) reports that 16,183 doses of vaccine have been administered in the past 24 hours, of which 10,013 doses of Pfizer vaccine, 522 of Moderna vaccine, 217 of AstraZeneca vaccine and 5,431 of Johnson & Johnson vaccine.
EMA recommends heart inflamation warning on COVID-19 vaccines in very rare cases
In very rare cases following vaccination with the COVID-19 vaccinesComirnaty and Spikevax (previously COVID-19 Vaccine Moderna)
COVID-19: Pfizer Suggests Third Dose Booster against Delta Variant for Vaccinated
Pfizer is preparing a third booster Covid shot to take on the deadly Delta variant, the company revealed.
The pharma corporation is seeking emergency authorization from the US FDA for another jab to be administered six-to-twelve months after full vaccination.
Pfizer pushes for 3rd shot as variant drives global outbreaks
Pfizer and BioNTech announced on July 8 they would seek authorization for a third dose of their COVID-19 vaccine to boost its efficacy, as the Delta variant drove devastating outbreaks in Asia and Africa and cases rose again in Europe and the United States.
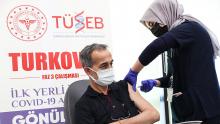
Turkey holds Phase 3 trials for local vaccine candidate
Turkey has begun to administer its national COVID-19 vaccine candidate, TURKOVAC, on volunteers in Istanbul as part of Phase 3 trials.
Jointly developed by the Health Ministry, Health Institutes of Turkey (TUSEB) and Erciyes University, the national inactive candidate vaccine is being administered at the Başakşehir Çam and Sakura City Hospital ahead of wider use.
COVID-19 vaccination campaign: 16,311 people immunised in 24h, 9,938 with first dose
Romania's National COVID-19 Vaccination Coordination Committee (CNCAV) reports that 16,311 doses of vaccine have been administered in the past 24 hours, of which 9,918 doses of Pfizer vaccine, 552 of Moderna vaccine, 321 of AstraZeneca vaccine and 5,520 of Johnson & Johnson vaccine. CNCAV shows that 9,938 people were vaccinated with the first dose and 6,373 received the second dose.